 
When heated in air, thorium metal ignites and burns brilliantly with a white light. However, when it is contaminated with the oxide, thorium slowly tarnishes in air, becoming grey and eventually black.  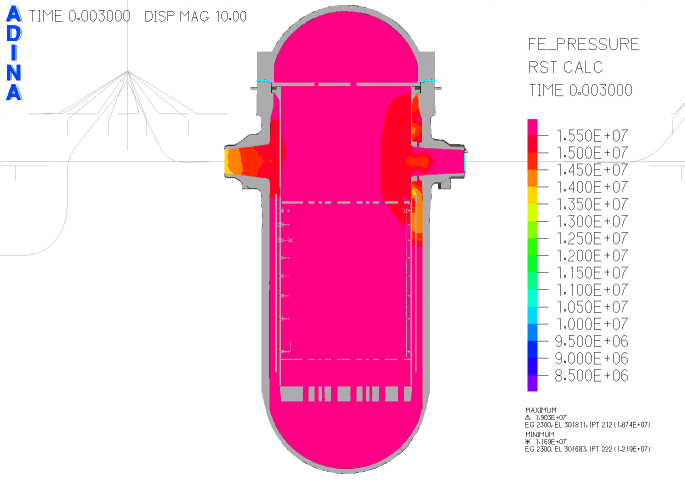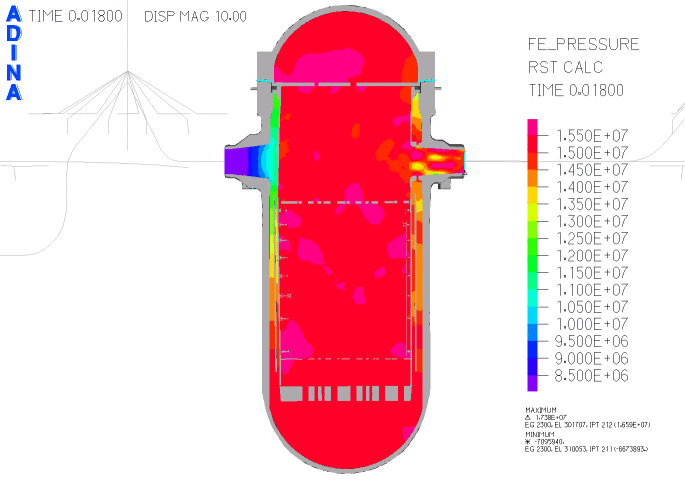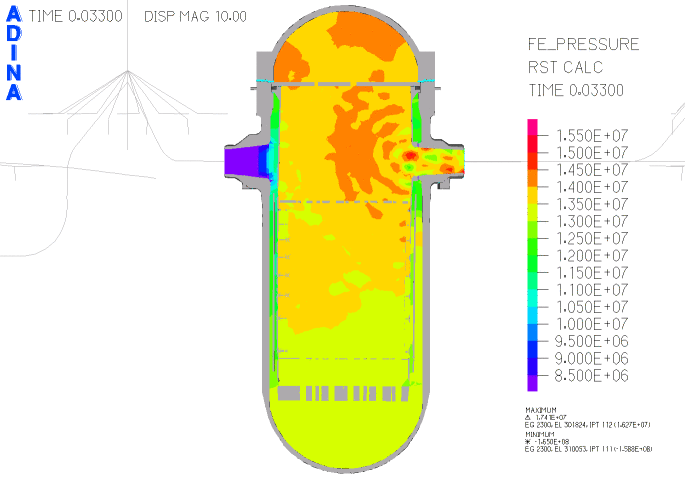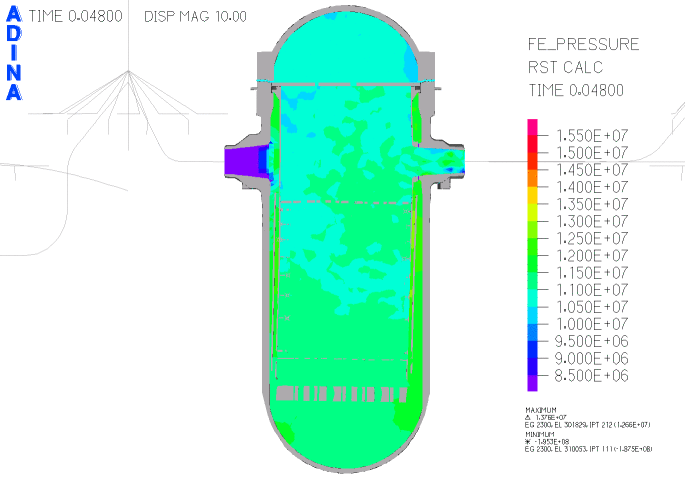
When pure, thorium is a silvery white metal that retains its lustre for several months. The decay chains of natural thorium and uranium give rise to minute traces of Th-228, Th-230 and Th-234, but the presence of these in mass terms is negligible. Thorium exists in nature in a single isotopic form – Th-232 – which decays very slowly (its half-life is about three times the age of the Earth). Soil contains an average of around 6 parts per million (ppm) of thorium. Thorium is very insoluble, which is why it is plentiful in sands but not in seawater, in contrast to uranium. It is found in small amounts in most rocks and soils, where it is about three times more abundant than uranium. Thorium is a naturally-occurring, slightly radioactive metal discovered in 1828 by the Swedish chemist Jons Jakob Berzelius, who named it after Thor, the Norse god of thunder. This is occurring preeminently in China, with modest US support. Extracting its latent energy value in a cost-effective manner remains a challenge, and will require considerable R&D investment. The use of thorium as a new primary energy source has been a tantalizing prospect for many years. Molten salt reactors are well suited to thorium fuel, as normal fuel fabrication is avoided.Thorium fuels can breed fissile uranium-233 to be used in various kinds of nuclear reactors.It is fertile rather than fissile, and can only be used as a fuel in conjunction with a fissile material such as recycled plutonium.Thorium is more abundant in nature than uranium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
